Alcohol Hangovers: The Role of Methyl Alcohol

In my last blog, I discussed the issue of hangovers. While simple enough to get, hangovers are a complex phenomenon.
All alcoholic beverages consist of water, alcohol and varying concentrations and types of congeners. Congeners are chemicals which are produced during the fermentation, distillation and production of alcohol and include such chemicals as other fusel alcohols, such as methyl, isopropyl and butyl alcohols; aldehydes, and esters. Congeners contribute to the unique taste and aroma of the alcoholic beverage.
Congeners, which tend to be in higher concentrations in distilled liquors, are not clear and have been implicated in hangovers. The odds ratio for a hangover was 1.41X greater than that of vodka when brandy was consumed, and was 0.85X when beer was consumed (WOA60801). One congener that has been particularly associated with hangovers is methyl alcohol. A detailed review study on congeners has just been published as follows:
Rodda, L.N., Beyer, J., Gerostamoulos, and Drummer, O.H., “Alcohol Congener Analysis and the Source of Alcohol: A Review”, Forensic Science, Medicine and Pathology, 9: 194-207, 2013 (WOA608U2)
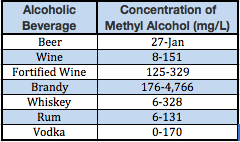
The methyl alcohol concentration of various alcoholic beverages from that study is listed in the following Table.
Methyl Alcohol is Naturally Produced in the Gut
Methyl alcohol is also produced naturally in the gut by bacteria, especially when various types of fruit containing high amounts of pectin are eaten. Methyl alcohol produced by the body is eliminated mainly by enzymes in the liver – the same enzyme that metabolizes alcohol, as follows:
Methyl→Formaldehyde→Formic→Carbon dioxide + water alcohol acid
Methyl alcohol by itself is relatively non toxic, but the products such as formaldehyde and formic acid are approximately 6X more toxic and can cause symptoms similar to hangover. Normally methyl alcohol and its metabolites are in extremely low concentrations in the blood and do not cause any symptoms. The situation changes however, when alcohol is consumed.
Alcohol Inhibits the Metabolism of Methyl Alcohol
Alcohol prevents the liver enzymes from metabolizing methyl alcohol (as shown by the symbol ) and so the blood methyl alcohol concentration increases during drinking. When the blood alcohol concentration becomes less than 0.01 g/100mL, the enzyme system is no longer blocked and a large amount of formaldehyde and formic acid are now produced from the high blood methyl alcohol concentration which accumulated during the drinking.
The peak of methyl alcohol concentration in the urine coincides with the time when the hangover symptoms are the worse (WOA60804). So it would be expected that if one consumes alcoholic beverages such as vodka and clear rum that contain low amounts of methyl alcohol, that there would be no hangover. Unfortunately these drinks are usually mixed with fruit juices which contain pectin and are converted in the gut to methyl alcohol. As alcohol blocks the metabolism of methyl alcohol, the methyl alcohol concentration increases, potentially causing a greater incidence of hangover even in alcoholic beverages that are low in methyl alcohol concentrations.
Hangover Prevention
It goes without saying that the subject of hangover is a complex phenomenon. And, as you can probably surmise from these past two blogs, the only surefire way to prevent alcohol hangovers is by not drinking to excess.
In Memoriam – Constable John Zivcic
In light of recent news of the tragic death of Constable John Zivcic, I would like to extend my condolences to his family and the Toronto Police for the loss of this young police officer. It is reported that he was responding to a call of an impaired driver.
When I worked at the forensic lab in Toronto during the 1970’s and 1980’s, I always dreaded the holiday season. This was because, during December, there was a large increase in the number of postmortem blood and urine samples submitted from drinking drivers killed in motor vehicle collisions. It was, of course, very depressing to deal with this senseless loss of life especially during the festive celebrations. Fortunately, with increased media attention, anti drinking and driving ads, responsible office parties (in part due to social host liability laws), and especially the large increase and visibility of spot-checks and road-checks, there has been a dramatic decrease in road traffic deaths in December. For that reason, Constable Zivcic’s death is particularly poignant.
Where once December was the worst month, now December is one of the safer months. The worst months being from May to October. This is in no small part the result of the dedicated law enforcement personnel who give up time with their families to brave the dark winter nights conducting spot checks which have made the holiday season a safer season for us all. To them and in memory of Constable John Zivcic, I dedicate this blog.